43 jablonski diagram fluorescence
Fluorescence & Jablonski diagrams; Fluorescence & Jablonski diagrams. October 17, 2019 October 17, 2019. Welcome to the electron apartment building! ♀️ Would you like to take the elevator to a higher electronic state? Hold on, I've got to find a photon! I like to think of FLUORESCENCE as involving electrons moving between floors of a ... Fluorescence phenomenon represented in the Jablonski diagram (Llères et al., 2007) Because electrons will have different excited state levels and they will find different ground state levels ...
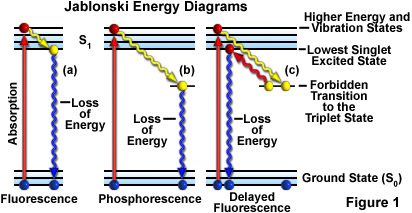
Fluorescence Excitation and Emission Fundamentals Fluorescence is a member of the ubiquitous luminescence family of processes in which ... by a Jablonski energy diagram (see Figure 1), named in honor of the Polish physicist Professor Alexander Jablonski. A typical Jablonski diagram illustrates the singlet ground (S(0)) state, as well
Jablonski diagram fluorescence
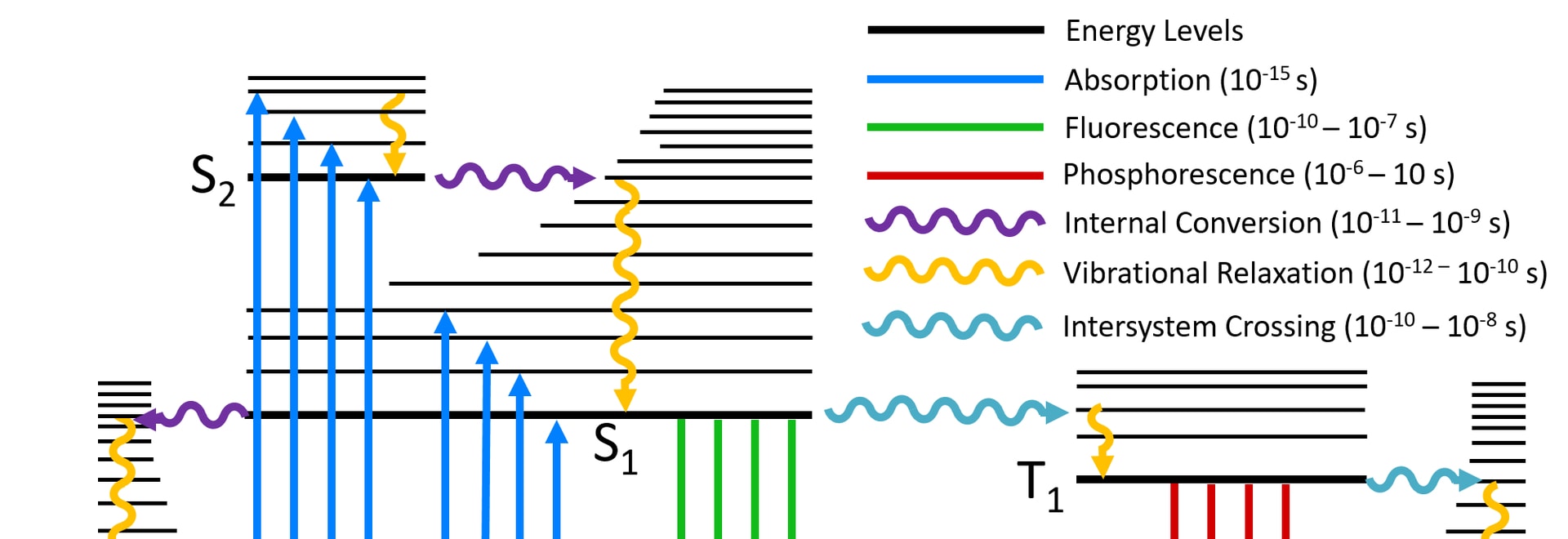
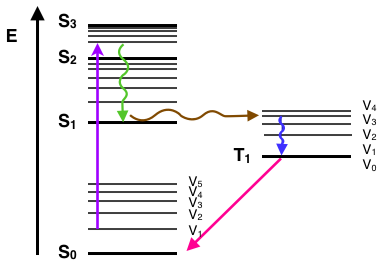
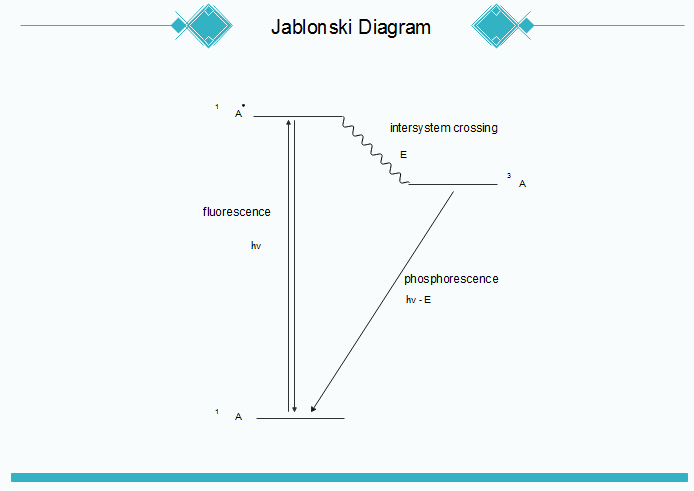
Jablonski diagram of FRET with typical timescales indicated. Förster (Fluorescence) resonance energy transfer (FRET), resonance energy transfer (RET) or electronic energy transfer (EET), is a mechanism describing energy transfer between two chromophores.[1] A donor chromophore, initially in its electronic Figure 5: Jablonski diagram of fluorescence and phosphorescence processes and their typical rate constants. Alternatively the molecule may undergo intersystem crossing (ISC) to the excited triplet state (T 1). ISC typically occurs in molecules with a high degree of spin-orbit coupling, the coupling of the orbital angular momentum and the spin ... Jablonski Energy Diagram. Fluorescence activity can be schematically illustrated with the classical Jablonski diagram, first proposed by Professor Alexander Jablonski in 1935 to describe absorption and emission of light. Prior to excitation, the electronic configuration of the molecule is described as being in the ground state.
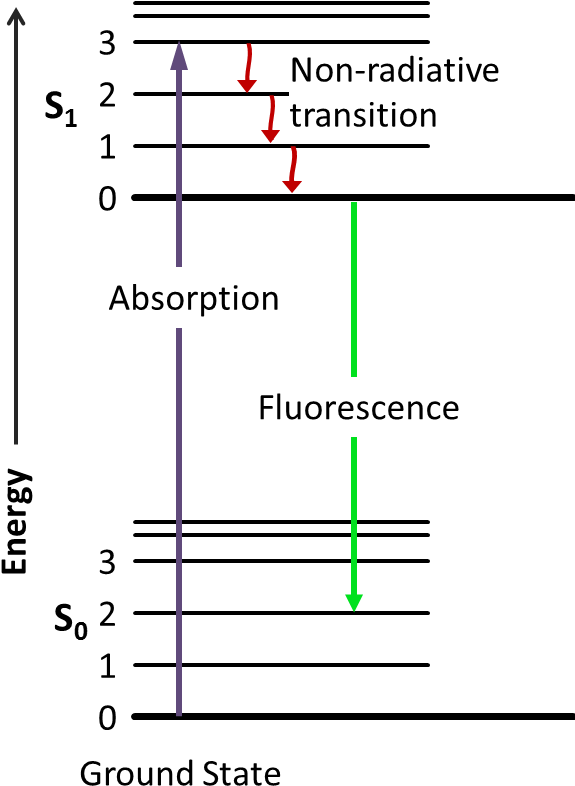
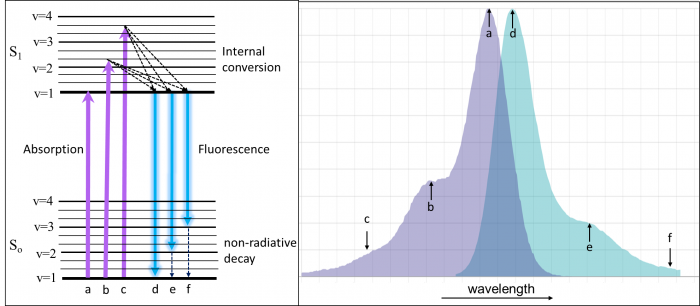
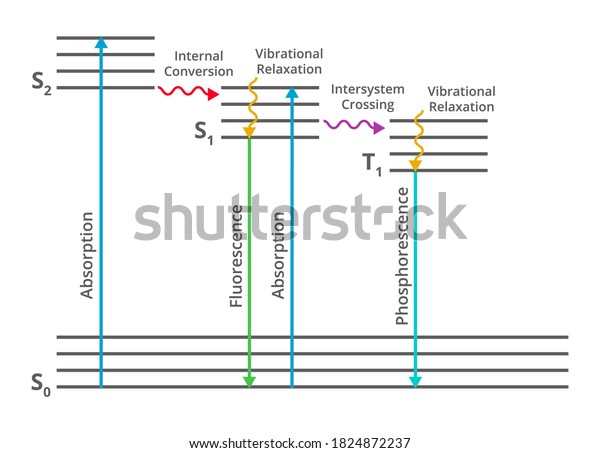
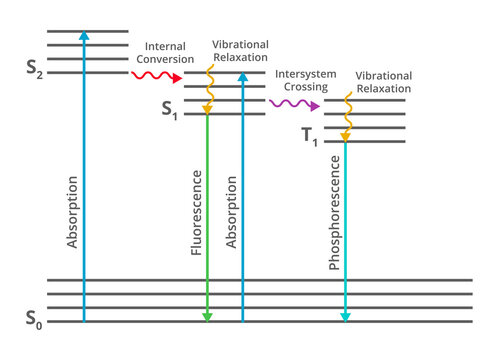
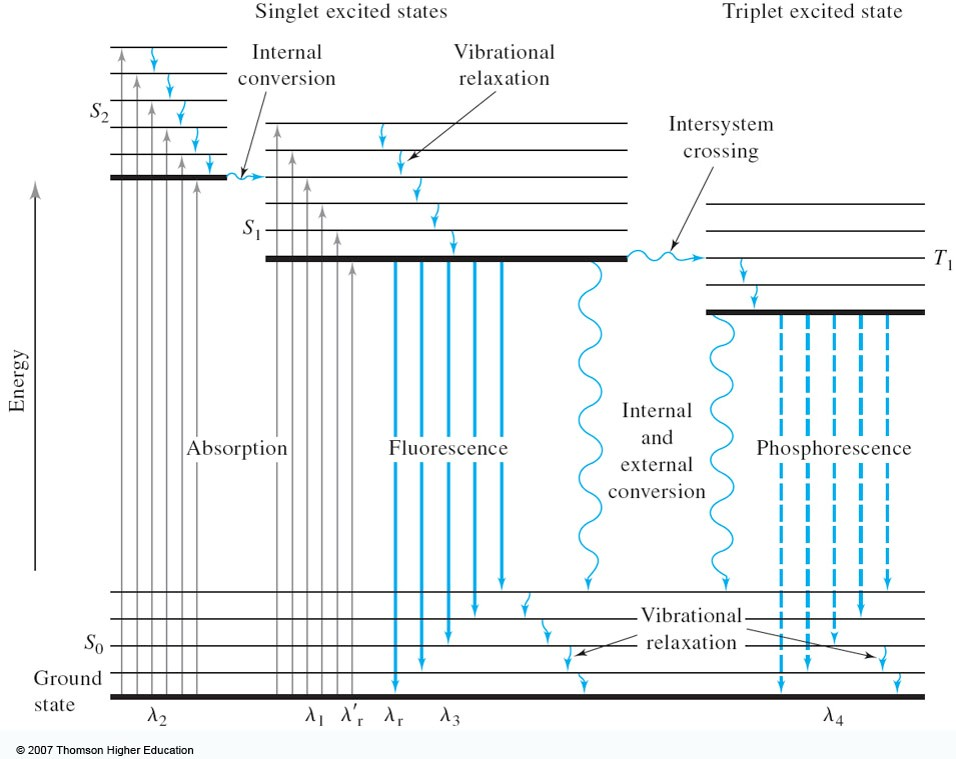
Jablonski diagram fluorescence. Whenever an electron moves to the excited state by the absorption of EMRs, there are several possibilities for relaxation of this electron which are explaine... Jablonski diagram including vibrational levels for absorbance, non-radiative decay, and fluorescence. Radiative transitions involve the absorption of a photon, if the transition occurs to a higher energy level, or the emission of a photon, for a transition to a lower level. Nonradiative transitions arise through several different mechanisms ... Jablonski diagram for fluorescence. An excited-state electron rapidly (on the order of 10 -12 seconds) loses its energy to vibration (heat), a process called internal conversion, and falls to the lowest level of the first (S 1) excited state. From there the electron may fall to one of the sub-levels of the ground (S 0) state, emitting a photon ... The diagram used to describe this process is called a Jablonski diagram, shown in Figure 1-5. Figure 1-5: Fluorescence, shown in this Jablonski diagram, involves emitting a photon at a lower energy than the photon that was initially absorbed. The diagram is read from left to right – absorbance occurs first, then
Jablonski Diagram - Java Tutorial. Fluorescence activity can be schematically illustrated with the classical Jablonski diagram, first proposed by Professor Alexander Jablonski in 1935 to describe absorption and emission of light. This tutorial explores how electrons in fluorophores are excited from the ground state into higher electronic energy ... The Jablonski diagram, typically used to illustrate fluorescence in molecular spectroscopy, demonstrates the excited states of a molecule along with the radiative and non-radiative transitions that can occur between them. Fig. 1 shows the Jablonski diagram (Jablonski, 1933), a schematic of the transition of electronic state of a molecule during ... A Jablonski diagram is basically an energy diagram, arranged with energy on a vertical axis. The energy levels can be quantitatively denoted, but most of these diagrams use energy levels schematically. The rest of the diagram is arranged into columns. Every column The Jablonski diagram, typically used to illustrate fluorescence in molecular spectroscopy, demonstrates the excited states of a molecule along with the ...
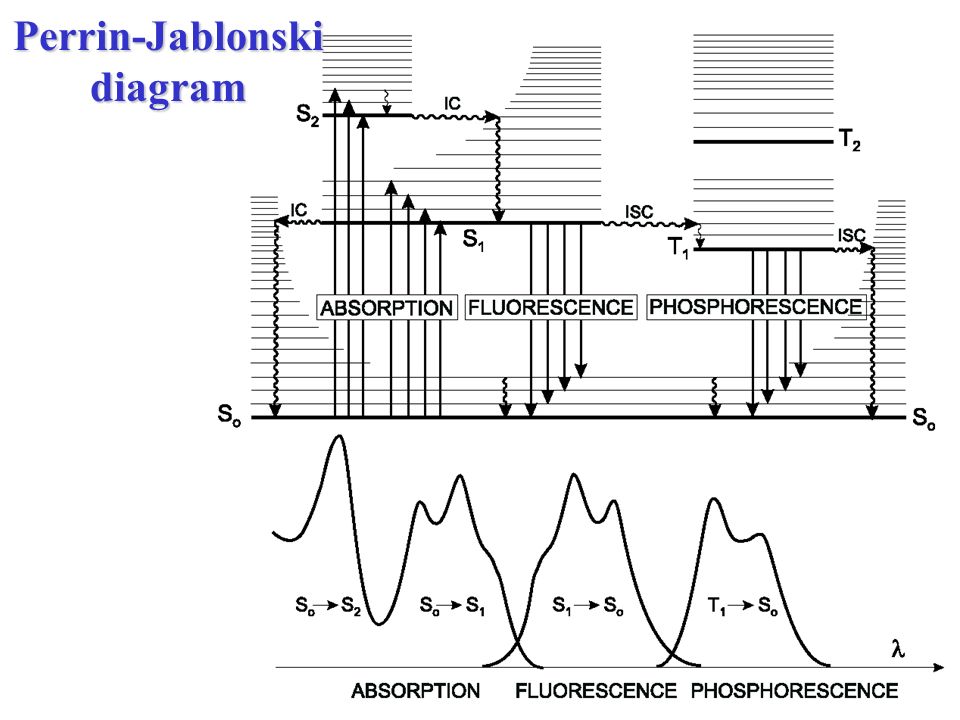
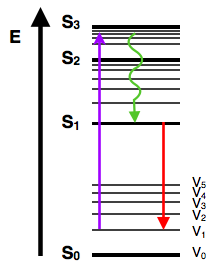
The Jablonski diagram, typically used to illustrate fluorescence in molecular spectroscopy, demonstrates the excited states of a molecule along with the radiative and non-radiative transitions that can occur between them. Fig. 1 shows the Jablonski diagram (Jablonski, 1933), a schematic of the transition of electronic state of a molecule during ... A fluorescent probe is a fluorophore designed to respond to a specific stimulus or to localize within a specific region of a biological specimen. The process responsible for the fluorescence of fluorescent probes and other fluorophores is illustrated by the simple electronic-state diagram (Jablonski diagram) shown in Figure 2. The Polish physicist Professor Aleksander Jablonski (1898-1980) was the first to describe the cycle between ground/excitation/emission in a three-level energy diagram which is known simply as a "Jablonski Diagram". In 1930, he received his PhD from Warsaw University for his thesis entitled "On the influence of the change of the wavelength of excitation light on the fluorescence spectra". Figure \(\PageIndex{2}\): Partial Jablonski Diagram for Absorption, Fluorescence, and Phosphorescence. from Bill Reusch. The upper lines represent the energy state of the three excited electronic states: S 1 and S 2 represent the electronic singlet state (left) and T 1 represents the first electronic triplet state (right).

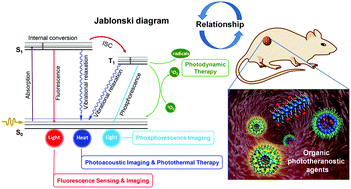
Design Of Superior Phototheranostic Agents Guided By Jablonski Diagrams Chemical Society Reviews Rsc Publishing
The Jablonski Diagram is named after Polish physicist Aleksander Jabłoński who, due to his many pioneering contributions, is regarded as the father of fluorescence spectroscopy. 1 He received his doctorate for the work “On the influence of the change of wavelengths of excitation light on the fluorescence spectra” where he provided ...
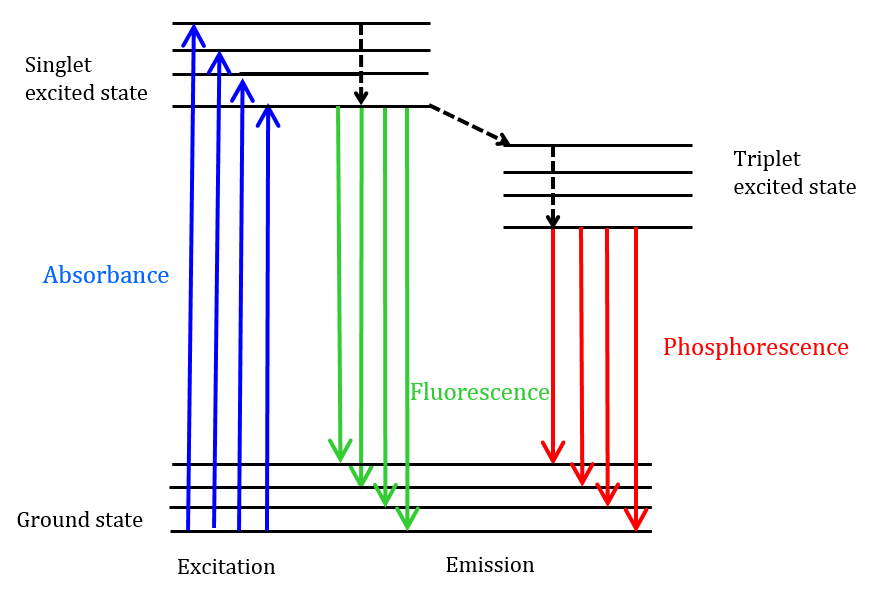
Fluorescence is a process that occurs after excitation of a molecule with light. It involves transitions of the outermost electrons between different electronic states of the molecule, resulting in emission of a photon of lower energy than the previously absorbed photon. This is represented in the Jablonski diagram (see Fig. 6.1).
Introduction to Fluorescence, and the Jablonski diagram
The Jablonski diagram was developed in 1933. It is a well respected schematic of the transition of the electronic state of a molecule during the phenomenon of fluorescence. In the diagram below, the left axis highlights increasing energy where a normal fluorescent molecule possesses an absorbance spectrum.
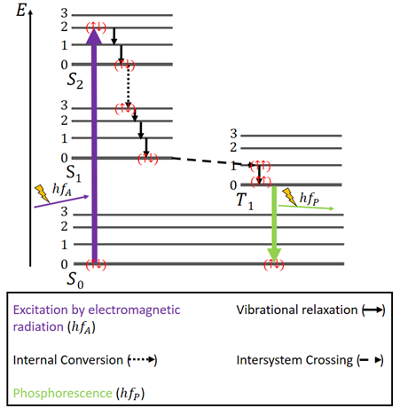
The Jablonski Diagram Explained . Flow cytometrists use the Jablonski diagram to aid in understanding and explaining the kinetic events of fluorescence.. Fluorescent compounds start at the ground state (S 0) until they are excited by interacting with a photon of light (Step 1). This photon excites the compound, promoting an electon to a higher energy state (S 1 ’).
A typical Jablonski diagram illustrates the singlet ground state (S(0)), in addition to the first (S(1)) and second (S(2)) excited singlet states as a stack of horizontal lines.In the tutorial, the thicker lines represent electronic energy levels, while the thinner lines denote the various vibrational energy states (rotational energy states are ignored).
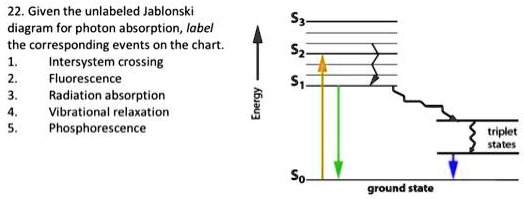
Solved Given The Unlabeled Jablonski Diagram For Photon Absorption Label The Corresponding Events On The Chart Intersystem Crossing Fluorescence Radiation Bsorption Vibrational Relaxation Phosphorescence Triplet Hel Ground State
Jablonski Diagram. Fluorescence activity can be schematically illustrated with the classical Jablonski diagram, first proposed by Professor Alexander Jablonski in 1935 to describe absorption and emission of light. This tutorial explores how electrons in fluorophores are excited from the ground state into higher electronic energy states and the ...
fluorescence phosphorescence Jablonski diagram fluorescence spectrum triplet state singlet state intersystem crossing dissociation laser stimulated emission gain population inversion four-level laser pumping photoejection photoelectrons photoelectron spectrum For a non-linear molecule containing N atoms:
Jablonski diagram: If the photon emission (shown here as a short wavelength, that is downward pointing, "long" green line in the diagram) occurs between states of the same spin state (e.g. S 1 ---> S 0) this is termed fluorescence. If the spin state of the initial and final energy levels are different (e.g. T 1 --> S 0 ), the emission (loss of ...
What Is A Jablonski Diagram Perrin Jablonski Diagram 2 Bain Square Livingston Eh54 7dq Uk Edinburgh Instruments
Fluorescence. Another pathway for molecules to deal with energy received from photons is to emit a photon. This is termed fluorescence. It is indicated on a Jablonski diagram as a straight line going down on the energy axis between electronic states.
Download Chemsummer Carnival Glow Sticks Jablonski Diagram Fluorescence Lifetime Png Image With No Background Pngkey Com
This phenomenon is well-known as fluorescence. Sometimes the light absorbed is given out slowly and even long after the removal of the source of light. This phenomenon is known as phosphorescence. The phenomena of fluorescence and phosphorescence are best explained with the help of the Jablonski Diagram.
NEWS AND VIEWS 401 At present, his diagram appears in most textbooks on photochemistry and photobiology; as he wrote in 1935: "by these deliberations the phenomena of photoluminescence have lost some of their mysterious character" [ 2 ] . 1 A, Jablonski, Efficiency of anti-Stokes fluorescence in dyes, Nature, 131 (1933) 839 - 840. 2 A ...
Jablonski Energy Diagram. Fluorescence activity can be schematically illustrated with the classical Jablonski diagram, first proposed by Professor Alexander Jablonski in 1935 to describe absorption and emission of light. Prior to excitation, the electronic configuration of the molecule is described as being in the ground state.

Ca2 Detection In Muscle Tissue Using Fluorescence Spectroscopy Surgical Instruments Research Instruments Laboratory Equipment Wpi
Figure 5: Jablonski diagram of fluorescence and phosphorescence processes and their typical rate constants. Alternatively the molecule may undergo intersystem crossing (ISC) to the excited triplet state (T 1). ISC typically occurs in molecules with a high degree of spin-orbit coupling, the coupling of the orbital angular momentum and the spin ...
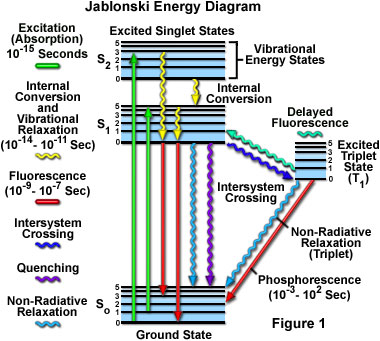
Molecular Expressions Microscopy Primer Fluorescence Jablonski Energy Diagram Interactive Java Tutorial
Jablonski diagram of FRET with typical timescales indicated. Förster (Fluorescence) resonance energy transfer (FRET), resonance energy transfer (RET) or electronic energy transfer (EET), is a mechanism describing energy transfer between two chromophores.[1] A donor chromophore, initially in its electronic

Chemsummer Carnival Glow Sticks Jablonski Diagram Fluorescence Hd Png Download Transparent Png Image Pngitem

See Figure Jablonski Diagram This Figure Describe The Absorbance Fluorescence Phosphorescence Internal From Publication Diagram Scientist Interactive

Solved 1 10 Points For A Given Jablonski Diagram Below Answer The Following Questions With Your Reasoning Based On The Course Material About Molecular Spectroscopy Singlet Excited Stales Vibrational Interal Relaxation Convcrsion Triplat

Jablonski Diagram Photophysical Processes Fluorescence Phosphorescence Radiative And Non Radiative Youtube
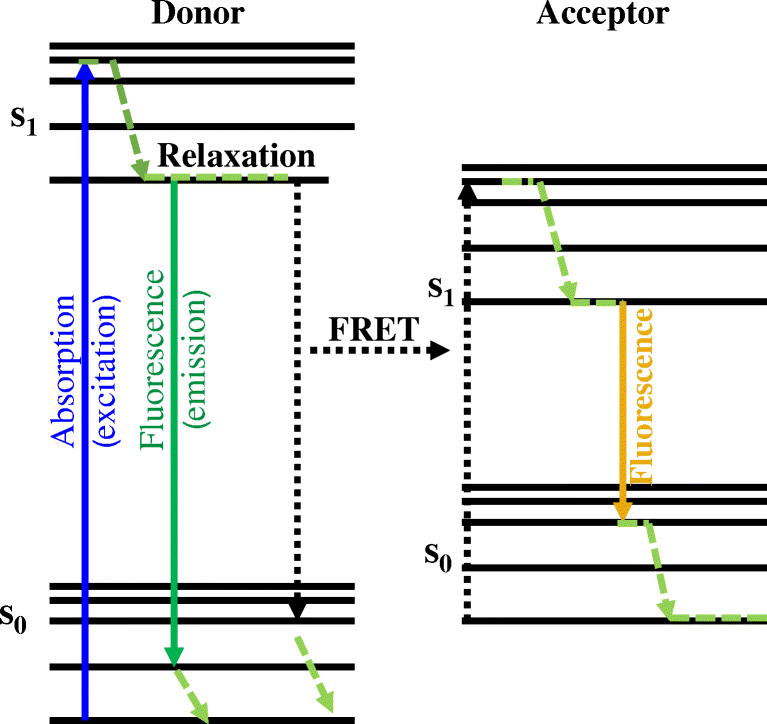
Figure 7 Optical Approaches For Single Cell And Subcellular Analysis Of Gpcr G Protein Signaling Springerlink


.jpg)







Comments
Post a Comment